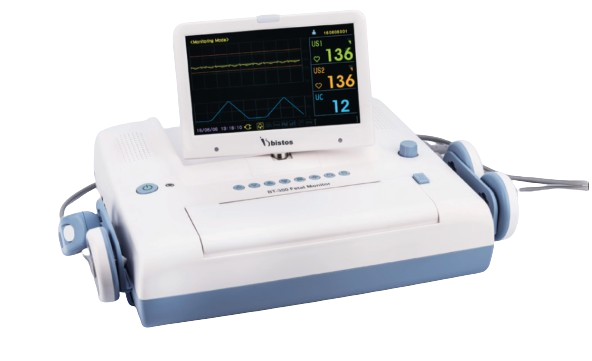
Bistos BT-350 Fetal Monitor
The Bistos BT-350 is a lightweight, portable fetal monitor providing FHR (30–240 bpm), maternal pulse (30–240 bpm), TOCOCONTRACTION (0–100%), and manual fetal movement detection with built-in thermal printer. Ideal for OB/GYN clinics and home care, it features ultrasound Doppler (1 MHz), 9-crystal TOCO transducer, rechargeable battery (8 hours), 2.4-inch LCD, and triplet monitoring option for reliable, non-invasive fetal well-being assessment.
Description
Design and Protection Mechanism: Portable Antepartum Configuration The Bistos BT-350 is a user-centric, compact fetal monitor tailored for antepartum surveillance in outpatient clinics, labor wards, and home-based maternal care, ensuring continuous or intermittent tracking of fetal heart rate (FHR) and uterine activity without invasive procedures. Constructed with a rugged ABS plastic casing and ergonomic handle for portability, it weighs just 1.5 kg, making it suitable for bedside or mobile use. The IPX1-rated transducers (ultrasound and TOCO) resist minor spills, while the rechargeable lithium battery provides up to 8 hours of operation with low-battery alerts. Safety interlocks include signal quality indicators to prevent false readings, and the device complies with IEC 60601-1 for electrical safety and ISO 13485 for quality in obstetric monitoring.
Performance and Signal Acquisition: Delivers accurate FHR detection (30–240 bpm, ±1 bpm resolution) via 1 MHz pulsed Doppler ultrasound with autocorrelation processing to minimize artifacts from maternal movement or fetal kicks. The TOCO channel measures uterine contractions (0–100% relative units) with high sensitivity for early labor detection, while manual fetal movement marking allows clinician annotation. The system supports NST (non-stress test) protocols with automatic FHR baseline calculation and variability assessment, achieving >99% accuracy in singleton pregnancies and optional triplet mode for multiples via additional transducers.
Transducer and Accessibility: Includes two wireless transducers (ultrasound belt-style and TOCO belt) with event markers for patient-activated movement counts, connecting via Bluetooth for cable-free mobility (up to 10 m range). The adjustable belly belt ensures secure placement on maternal abdomen, accommodating gestations from 20 weeks onward. The compact footprint (250 × 200 × 50 mm) fits on nightstands or carts, with a flip-open stand for tabletop stability and easy transducer docking.
User Interface and Operational Features: Features a 2.4-inch high-contrast LCD displaying real-time FHR waveforms, TOCO traces, and numerical values, with intuitive scroll-wheel navigation and multilingual menus (up to 6 languages, including English and Korean). Built-in thermal printer outputs 8 cm wide strips at 1 cm/min speed for 20–30 minute NSTs, with customizable formats (FHR/TOCO/MFM). The rechargeable battery supports 200+ print cycles, and USB port enables data transfer to PC for archiving or telehealth review.
Sterilization and Maintenance: Transducers wipe clean with 70% isopropyl alcohol or EPA disinfectants, with autoclavable gel pads optional. The sealed battery compartment and modular design allow easy transducer replacement; self-diagnostics at startup check signal integrity and battery health. Annual calibration via service port ensures accuracy (±2 bpm for FHR); low-maintenance LEDs last >10,000 hours, with a 2-year warranty on electronics.
Electrical and Environmental Specifications: Operates on built-in 7.4V Li-ion battery (charger included) or AC 100–240V adapter at <20W. Functions in 10–40°C ambient with ≤85% RH, with EMI shielding (IEC 60601-1-2) to avoid interference from mobiles or monitors. Low power mode extends battery life by 20% during intermittent use.
Dimensions and Portability: Ultra-portable at 250 × 200 × 50 mm (W × D × H) weighs 1.5 kg, with a carrying bag for transport. The flip stand and strap enable wall-mount or bedside use; setup in <1 minute with plug-and-play transducers.
- Throughput and Workflow: Monitors 5–10 patients daily with <2-minute setup, ideal for outpatient NSTs or home rentals.
- Detection and Monitoring: Doppler autocorrelation for FHR; TOCO piezoresistive sensor; logs 100+ sessions via USB.
- Detection Principle: 1 MHz pulsed-wave ultrasound with signal processing for FHR/uterine activity.
- Additional Features: Triplet capability; telehealth export; event marker button; low-signal alarm.
- Data Storage: 50 NST traces (up to 60 min each) printable or exportable in PDF.
- Application Modes: Antepartum NST, home monitoring, multiples; compatible with OB software.