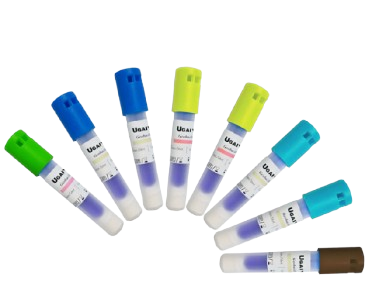
Fullcare FCB10301 Biological Indicator
The Fullcare FCB10301 is a self-contained biological indicator for steam sterilization validation, containing Geobacillus stearothermophilus spores (10^6 population) with built-in ampoule and growth medium. Ideal for autoclaves in hospitals, labs, and pharma facilities, it features 7-day incubation at 60°C, fluorescent color change readout, no biological waste, and ISO 11138 compliance for accurate, user-friendly efficacy confirmation.
Description
Design and Protection Mechanism: Self-Contained Ampoule Configuration The Fullcare FCB10301 is a compact, single-use biological indicator designed for precise validation of steam sterilization cycles in autoclaves, gravity, or vacuum processes, ensuring the elimination of microbial contaminants in healthcare, laboratory, and pharmaceutical settings. Encased in a polypropylene vial with a heat-sealed filter paper strip inoculated with Geobacillus stearothermophilus spores (ATCC 7953, population 1.0 x 10^6), it integrates a crushable glass ampoule of nutrient medium for post-exposure incubation. The tamper-evident cap and labeled exterior prevent contamination, while the robust construction withstands 121–134°C exposure without rupture; compliant with ISO 11138-3 and AAMI ST79 standards, it provides a direct lethality measure for steam processes (F0 >8 minutes) without generating hazardous biological waste.
Performance and Spore Resistance: Exhibits high resistance to steam sterilization with a D121 value of 1.5–2.5 minutes and z-value of 9–10°C, ensuring it survives sub-lethal cycles while confirming lethality in validated processes. Post-exposure, the ampoule is crushed to release medium, incubating at 60°C (±0.5°C) for 7 days (or 48 hours accelerated option) to detect survivors via metabolic byproducts. The fluorescent pH indicator shifts from yellow to red/orange if growth occurs, achieving >99.9999% log reduction detection with false-positive rate <0.1%; validated for 121°C/15 min, 132°C/4 min, and 134°C/3 min cycles.
Indicator and Accessibility: The self-contained design simplifies handling—no separate vials or transfer needed—fitting standard autoclave pouches or cassettes for easy loading. The ampoule’s ergonomic shape allows one-handed crushing post-cycle, and the color-coded label (green for viable, red for non-viable) provides immediate visual confirmation. Packaged in 10-unit trays for batch testing, it supports routine load release, weekly Bowie-Dick tests, or quarterly efficacy checks, with accessibility for gloved users in sterile environments.
User Interface and Operational Features: Includes pre-printed labels with lot number, expiration (2-year shelf life at 2–25°C), and QR code for traceability via app scan. The incubation readout is binary (growth/no growth) with optional fluorescent reader for quantitative fluorescence (excitation 365 nm, emission 460 nm), supporting up to 50 indicators per run. Multilingual instructions (English, Chinese, Spanish) and compatibility with incubators or readers streamline workflows, with no power required for the indicator itself.
Sterilization and Maintenance: The polypropylene vial withstands ethylene oxide or gamma irradiation for initial sterilization, with no maintenance needed beyond storage in dry, dark conditions. Post-use, non-growth indicators are disposed as regular waste, while growth-positive ones are autoclaved again; the ampoule’s one-time use eliminates reprocessing risks. Lot-specific certificates ensure traceability, with shelf-life testing per ISO 11138 for consistent spore viability.
Environmental Specifications: Stable at 2–25°C storage with <70% RH, the indicator tolerates -20–60°C transport without degradation. Non-toxic components (nutrient broth, spore suspension) pose no environmental hazards, and the compact size (50 × 10 mm vial) minimizes packaging waste.
Dimensions and Portability: Vial measures 50 × 10 mm, with tray packaging of 100 × 80 × 20 mm for 10 units, weighing 0.05 kg per indicator. Easily portable in lab kits or autoclave loads, with no special handling beyond standard biohazard precautions for positives.
- Throughput and Workflow: Validates 10–50 loads per batch with 7-day readout, supporting daily/weekly cycles in high-volume sterilizers.
- Detection and Monitoring: Spore growth via pH-fluorescent shift; optional reader for 0.1 log precision.
- Detection Principle: Geobacillus stearothermophilus spore survival post-steam exposure, with metabolic indicator.
- Additional Features: QR traceability; accelerated 48-hour option; Bowie-Dick compatible.
- Data Storage: Lot certificates; app logs for digital records.
- Application Modes: Steam autoclave validation, load release, efficacy testing; suitable for pharma/medical devices.